Atomistic ensemble of active SHP2
Abstract
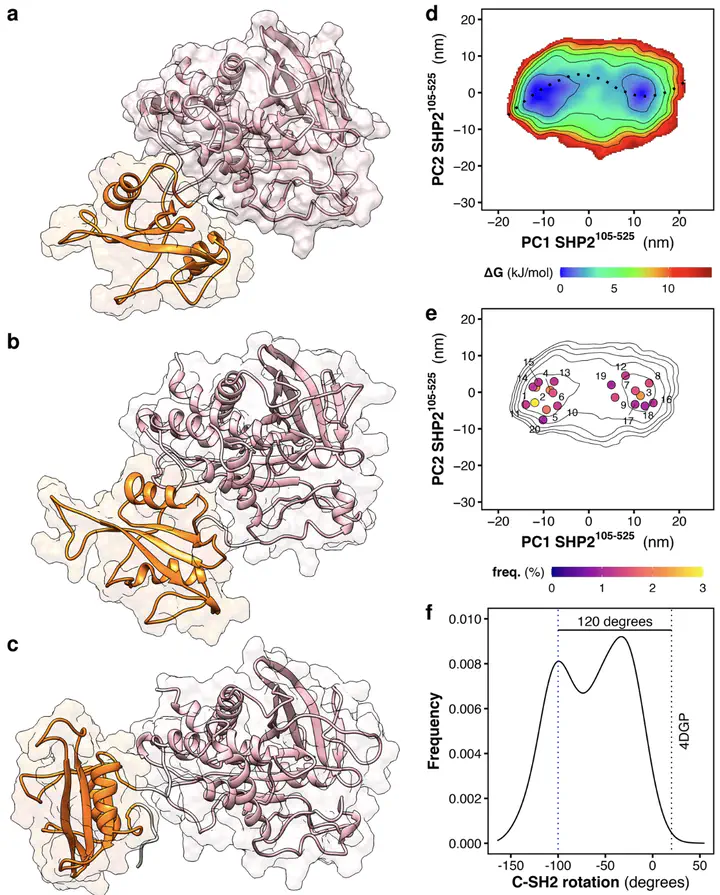
SHP2 phosphatase plays an important role in regulating several intracellular signaling pathways. Pathogenic mutations of SHP2 cause developmental disorders and are linked to hematological malignancies and cancer. SHP2 comprises two tandemly-arranged SH2 domains, a catalytic PTP domain, and a disordered C-terminal tail. In physiological, non-stimulated conditions, the catalytic site of PTP is occluded by the N-SH2 domain, so that the basal activity of SHP2 is low. Whereas the autoinhibited structure of SHP2 has been known for two decades, its active, open structure still represents a conundrum. Since the oncogenic mutant SHP2E76K almost completely populates the active, open state, this mutant has been extensively studied as a model for activated SHP2. By molecular dynamics simulations and accurate explicit-solvent SAXS curve predictions, we present the heterogeneous atomistic ensemble of the constitutively active SHP2E76K in solution, encompassing a set of conformational arrangements and radii of gyration in agreement with experimental SAXS data.