Abstract
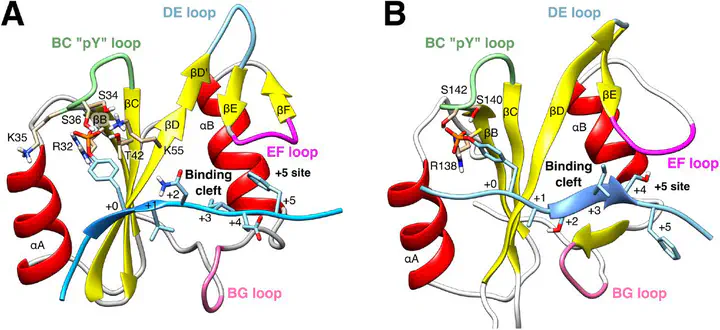
Src-homology 2 (SH2) domains are protein interaction domains that bind to specific peptide motifs containing phosphotyrosine. SHP2, a tyrosine phosphatase encoded by PTPN11 gene, which has been emerged as positive or negative modulator in multiple signaling pathways, contains two SH2 domains, respectively, called N-SH2 and C-SH2. These domains play a relevant role in regulating SHP2 activity, either by recognizing its binding partners or by blocking its catalytic site. Considering the multiple functions that these domains carry out in SHP2, N-SH2 and C-SH2 represent an interesting case of study. In this chapter, we present a methodology that permits, by means of the principal component analysis (PCA), to study and to rationalize the structures adopted by the SH2 domains, in terms of the conformations of their binding sites. The structures can be distinguished, grouped, classified, and reported in a diagram. This approach permits to identify the accessible conformations of the SH2 domains in different binding conditions and to eventually reveal allosteric interactions. The method further reveals that the conformation dynamics of N-SH2 and C-SH2 strongly differ, which likely reflects their distinct functional roles.