Spontaneous Quaternary and Tertiary T-R Transitions of Human Hemoglobin in Molecular Dynamics Simulation
Abstract
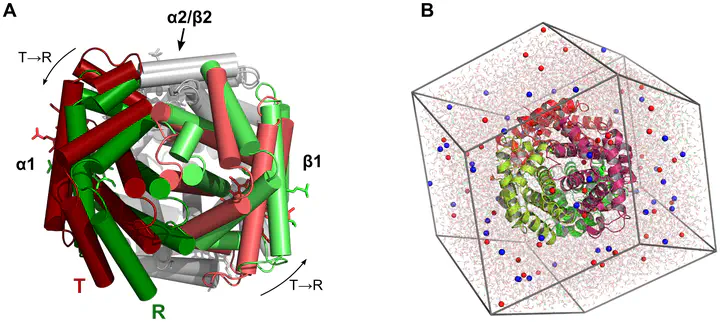
We present molecular dynamics simulations of unliganded human hemoglobin (Hb) A under physiological conditions, starting from the R, R2, and T state. The simulations were carried out with protonated and deprotonated HC3 histidines His(β)146, and they sum up to a total length of 5.6µs. We observe spontaneous and reproducible T $\rightarrow$ R quaternary transitions of the Hb tetramer and tertiary transitions of the α and β subunits, as detected from principal component projections, from an RMSD measure, and from rigid body rotation analysis. The simulations reveal a marked asymmetry between the α and β subunits. Using the mutual information as correlation measure, we find that the β subunits are substantially more strongly linked to the quaternary transition than the α subunits. In addition, the tertiary populations of the α and β subunits differ substantially, with the β subunits showing a tendency towards R, and the α subunits showing a tendency towards T. Based on the simulation results, we present a transition pathway for coupled quaternary and tertiary transitions between the R and T conformations of Hb.